

On the evening of July 24, 2023, Beijing time, Prof. Wu Yilong’s team published the research findings of Arm Group 7 (HER2-arm) of a large-scale umbrella trial (CTONG1702) and some data from a concurrent observational real-world study (CTONG 1705) in the top-tier international medical journal Nature Medicine (IF: 82.9). This study proposes a new clinical trial model for sufferers with rare genetic variations for the first time, and creates a clinical research design method that is more efficient and comprehensive in reflecting drug efficacy, facilitating the comprehensive collection of sufferer research data and the exploration of the most reasonable treatment plans.

Findings were published in a top-tier journal.
Clinical conflict: high inclusion threshold for clinical trials and unmet clinical needs for rare targets
In the age of biomarker-driven treatment, due to the low incidence of rare mutations and the limited number of sufferers carrying such genetic variations, the R&D cycles of targeted drugs are extended greatly, making it impossible for sufferers to accept potentially efficacious targeted drugs for a long time. Clinical trials are a valuable opportunity for these sufferers to come into contact with the most frontier drugs. According to statistics, only 44.8% of cancer sufferers have an opportunity to participate in clinical trials, while nearly half (21.9%) of them are excluded due to such constraints as inclusion criteria. Although strict inclusion criteria ensure the homogeneity of subjects, overly stringent conditions not only deprive some rare mutation sufferers of access to frontier investigational drugs but also often fail to represent the real-world efficacy and safety of such sufferers.
Design innovation: implementing a “sufferer-centered” innovative clinical trial model
“This is a research-centered clinical trial, rather than a sufferer-centered one. Considering long R&D cycles of new anti-cancer drugs, advanced stage sufferers who are not included are likely to have tragic outcomes.” said Prof. Wu Yilong. Therefore, in July 2018, Prof. Wu Yilong took the lead in starting a “sufferer-centered” national multicenter study (CTONG1702 and CTONG1705) providently. This study detected tumor tissues centrally through the second-generation sequencing platform of the central laboratory to concentrate on NSCLC sufferers with rare genetic variations. sufferers who met the strict inclusion criteria of the CTONG1702 study entered the “strict inclusion cohort”, while sufferers carrying rare targets but not meeting the inclusion criteria received medication treatment through compassionate drug use and entered the “compassionate drug use cohort”. Sufferers unwilling to be included in the CTONG1702 study but carrying rare targets for various reasons entered the observational real-world study CTONG1705 (the real-world cohort) for routine clinical treatment. Thus, by expanding the inclusion criteria, excluded sufferers also have an opportunity to receive frontier drug treatment, generating more efficacy and safety data from broader sufferer groups for medication treatment research. In addition, clinical outcomes of sufferers receiving real-world conventional clinical treatment are also observed, providing information on drug efficacy and safety to broader sufferer groups.
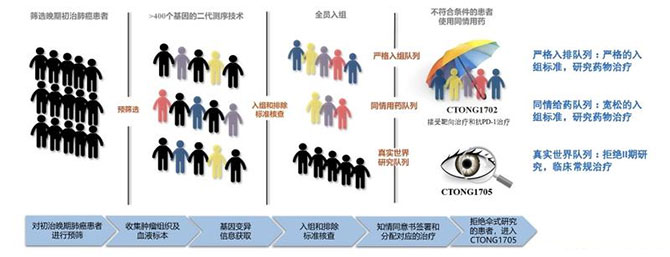
Illustration of study design
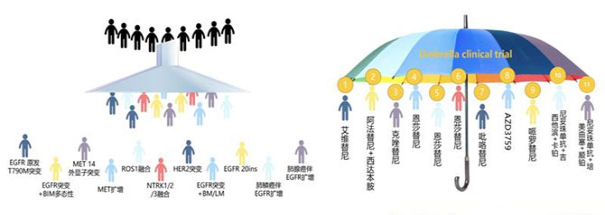
Illustration of umbrella trial
This publication mainly reports on the efficacy and safety of pyrotinib administered to HER2 mutant advanced NSCLC sufferers on the frontline at initial treatment, and the clinical outcomes of the real-world cohort. Prof. Wu Yilong said, “The number of cases in this study is limited, and the drug efficacy is not surprising. It was chosen and published by Nature Medicine, a top-tier journal ranking 11th globally, entirely due to the novelty of the study design. This is the first study in which sufferers are included in the same period, and which can demonstrate the efficacy and safety of a drug on the same target comprehensively without direct comparison. It also enables an indirect comparison with sufferers not receiving precise targeted treatment, thus providing a case for future clinical research on rare targets, and a new perspective for assessing an investigational drug comprehensively. As such, it holds high prospects and practical value.”
Applying the current sufferer-centered study design not only offers sufferers with rare genetic variations more opportunities to receive diverse new therapies but also increases the universality of investigational drugs, including pyrotinib, in broader groups. Similar study design models (e.g. CTONG2203 for ALK fusion) are also actively being prepared at Guangdong Provincial People’s Hospital.

The first authors of this article are postdoctoral fellow Liu Siyang from the First Affiliated Hospital of Jinan University, chief physician Tu Haiyan, and Dr. Wei Xuewu from the Guangdong Lung Cancer Institute, Guangdong Provincial People’s Hospital. The corresponding authors are Prof. Wu Yilong and Prof. Zhou Qing from the Guangdong Lung Cancer Institute, Guangdong Provincial People’s Hospital, and Prof. Li Yangqiu from Jinan University.
Guangdong Lung Research Institute
Updated: July 25, 2023